iRES Implant Systems Overview
 |
|
Shape One
|
|
|
|

|
|
iMax NHSI
|
|
|
|

|
|
iMax NHSIC
|
|
|
|

|
|
Volution
|
|
|
|

|
|
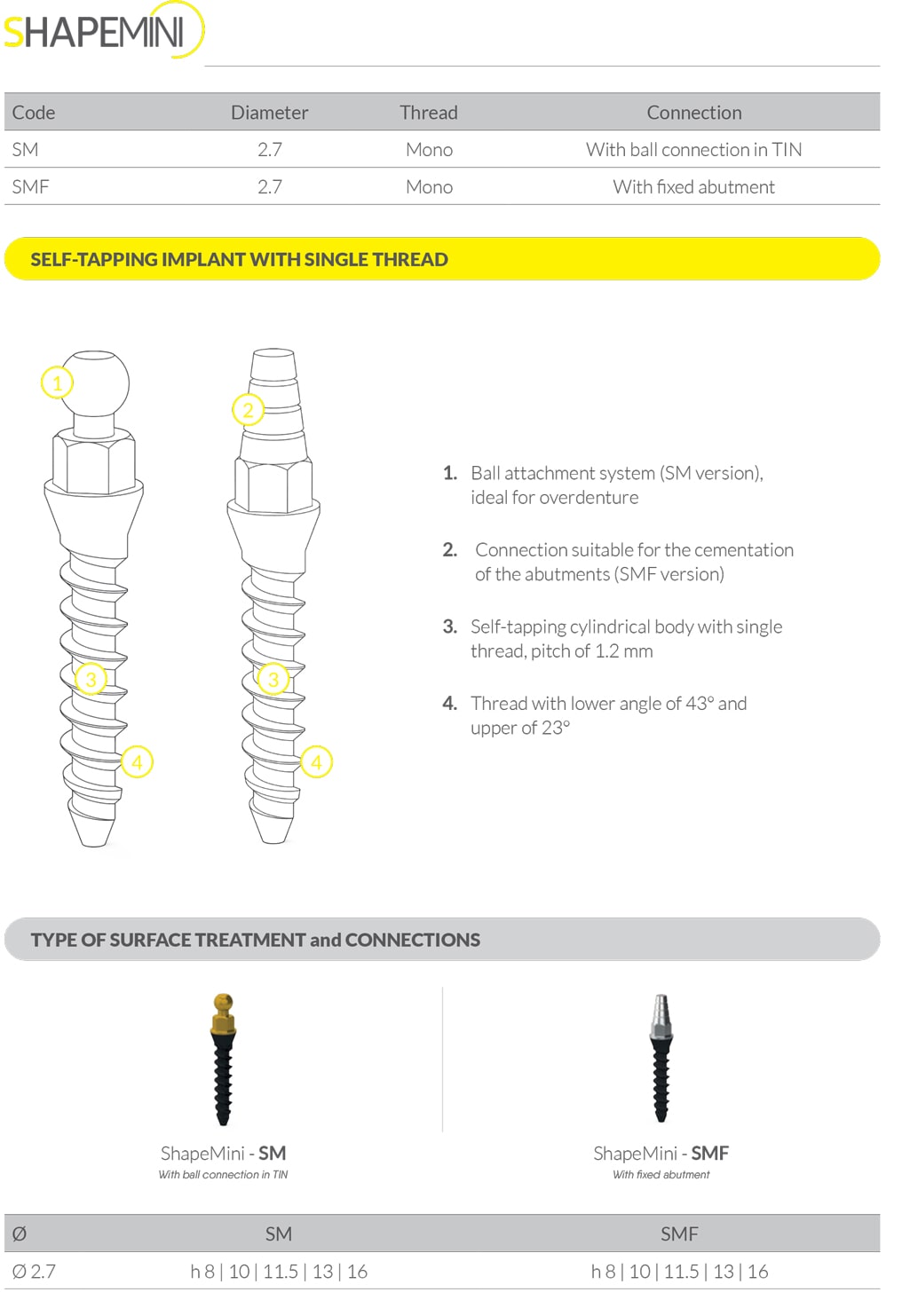
Shape Mini
|
|
|
|

|
|
|
IMPLANT CONNECTIONS
Prosthetic components with Friction Fit connection and one conical degree on the wall of the hex abutment, have been developed for S1B, S1TN, SVB and NHSI implant systems with internal hex connection. This connection ensures a “cold fusion” between implant and abutment if the retaining screw has been tightened at 30 Ncm. It eliminates micro-movements and reduces bacterial infiltration between implant and abutment. Prosthetic components with a tilted cone to 16° fit perfectly with our S1T implant system. An external hex connection with “Flat to Flat” joint has been developed for NHSE implant system. iRES® offers different implant solutions to cover a wide range of treatment options and fulfil every surgical need. The NHSIC conical connection has a 5° angle on the abutment and on the implant, and an emergence profile for the mucous attack. The cone-morse connection creates fissures (1µm) smaller than bacteria, absorbs vibration and chewing stress with the result that it eliminates the unscrewing of the screws. There is no connection between implant and abutment in NHSM Onepiece system and this allows to completely eliminate bacterial infiltration. OnePiece connection is perfect for immediate loading.



There is extensive scientific literature on how surface roughness characteristics influence cells behaviour. Compared to a smooth surface, topographical patterns smaller in size than a fibroblast cell (micro and nano topography) orient the arrangement of the cells and stimulate osteoblastic and platelet activity, accelerating the production of extracellular matrix and bone regeneration, and therefore the osseointegration of the dental implant. The three fundamentals of surface treatment of dental implants from a biological point of view are:
• Control of surface topography to stimulate a cellular response in an osteogenic direction;
• Control of the chemical composition of the surface to promote cell colonization;
• Control of biological contamination from adherent endotoxins so as not to interfere with the natural inflammatory response;
For the surface treatment a sand-blasting process was used, followed by a double acid attack. In the images, increasing the magnification, it is possible to observe how the macroscopic aspects of the screw (spire, cutting edge) are not affected by the treatment and how the surface is free from processing residue. The dual-beam roughness typical of SLA treatment can be observed and it contains large cavities due to large grit blasting on which is superimposed the micro-roughness due to treatment with acids. The micro-roughness illustrated in the figures highlights the typical three-dimensional topography, which gives these surfaces “sponge-like” characteristics that are the basis of their excellent clinical performance In fact, the very short peak-to-peak distance, about 1 micrometer, stimulates both the activity of osteogenic cells and the capillary penetration of the blood in the surface structure, offering very favorable characteristics to stimulate bone regeneration, as described in many articles on this topic. This unique combination of long-range roughness (large grit sand-blasting) and short-range (acid etching) is a substrate favourable to cell regrowth that adequately promotes cell differentiation. The level of roughness is Ra 1.42 ± 0.12.
Also hyaluronic acid enhances proliferation and growth of hydroxyapatite crystals.