-
منوی اصلیبستن
-
Dental
-
-
-
Gloves
-
-
-
Gloves
-
-
-
Gloves
-
-
-
Gloves
-
-
-
Dental Finishing & Polishing
-
-
-
Gloves
-
-
-
Gloves
-
-
-
Gloves
-
-
-
Gloves
-
-
-
Gloves
-
-
-
Gloves
-
-
-
-
Gloves
-
-
-
Gloves
-
-
-
Gloves
-
-
-
-
-
Gloves
-
-
-
Gloves
-
-
-
Gloves
-
-
-
Gloves
-
-
-
Gloves
-
-
-
Gloves
-
-
-
Gloves
-
-
-
- Dandal Service
- Dandal Yar
- Mag
- Help
-
Corporate
-
- Login/ Register
-
منوی اصلیبستن
-
Dental
-
-
-
Gloves
-
-
-
Gloves
-
-
-
Gloves
-
-
-
Gloves
-
-
-
Dental Finishing & Polishing
-
-
-
Gloves
-
-
-
Gloves
-
-
-
Gloves
-
-
-
Gloves
-
-
-
Gloves
-
-
-
Gloves
-
-
-
-
Gloves
-
-
-
Gloves
-
-
-
Gloves
-
-
-
-
-
Gloves
-
-
-
Gloves
-
-
-
Gloves
-
-
-
Gloves
-
-
-
Gloves
-
-
-
Gloves
-
-
-
Gloves
-
-
-
- Dandal Service
- Dandal Yar
- Mag
- Help
-
Corporate
-
- Login/ Register
- Home
- Medical & Surgical Equipment
- Medical & Surgical Supplies
- Cardiology
- CARDIOVASCULAR SURGERY
- Heart Valve
- Medtronic - Hancock II Valve
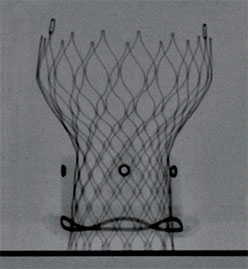
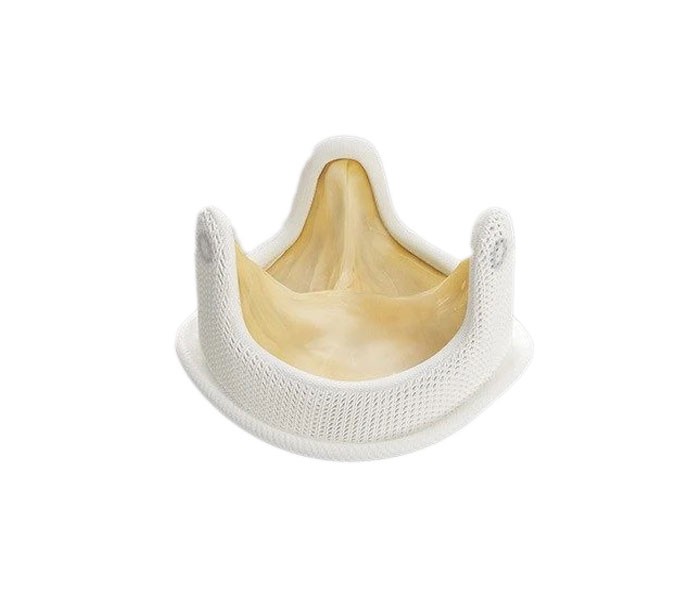
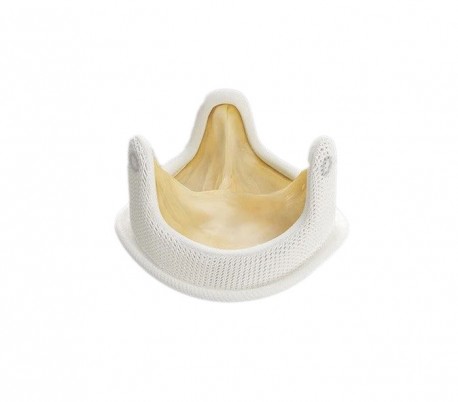
Medtronic - Hancock II Valve
-
In line with the demand of Dandal site customers for credit purchases, Dandal in cooperation with Saman Bank has provided a new service for dental and medical offices and treatment centers, which customers can use a credit card from the site at any time without any cash support.
- More Info
Medtronic - Hancock II Valve
Out-of-StockFeatures:
USA Made Biological Heart Valve
- Out stock
- Free
Pay in installments
Choose your plan:
Current plan: 3Month
Description: 30+3
Down payment: تومان0 (25.00%)
Number of payments: 3
Tax: تومان0
Amount of payment: تومان0
Overpayment: تومان0
Total: تومان0
Current plan: Cash
Description: نقدی
Down payment: تومان0 (0.00%)
Number of payments: 1
Tax: تومان0
Amount of payment: تومان0
Overpayment: تومان0
Total: تومان0
Hancock II Valve
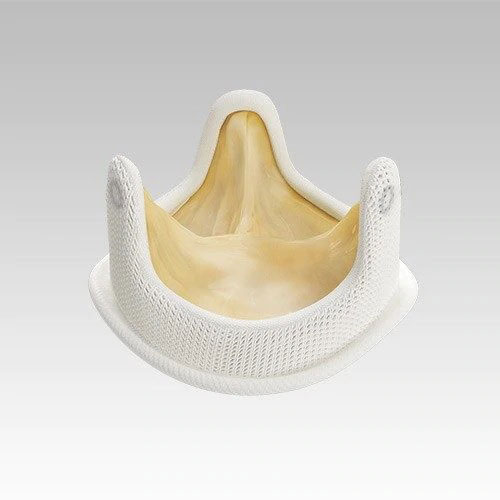
The Hancock II valve has stood the test of time. First introduced in the 1980s, it has been tested, studied and earned the trust of cardiac surgeons around the world. After dozens of years, and improvements along the way, there are scores of reasons to use this valve with your patients.
INDICATIONS
The Hancock II and Hancock II Ultra are for patients who require replacement of their native or prosthetic aortic and/or mitral valves.
PRODUCT DETAILS
TESTED, TRUSTED.
More than 30 years delivering consistent performance and clinical results, you can feel confident procedure after procedure.

DURABILITY
Published clinical experience demonstrates impressive long-term performance in all age groups for both the aortic and mitral valve.
Additional factors that may contribute to durability are the following:
- T6 (sodium dodecyl sulfate) anti-calcification treatment is applied in order to mitigate the absorption of calcium in the leaflets
- Next generation flexible polymer stent allows for absorption of stress produced during the cardiac cycle
- Low pressure fixation process minimizes septal muscle shelf allowing for improved hemodynamics
IMPLANTABILITY
Our innovative Cinch implant system further capitalizes on the valve’s flexible stent to facilitate valve implantation, particularly through a tight sinotubular space. It also:
- Improves overall visualization
- Has stent posts that deflect to allow for easier knot tying near the posts in aortic replacements
- Helps prevent suture looping
- Facilitates minimally invasive procedures
- Protects tissue from inadvertent damage
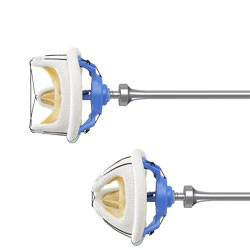
MAXIMUM FLOW
The Hancock II valve is designed to allow the maximum amount of blood to flow through it.
- The sewing ring is mounted flush with the inflow edge of the scalloped stent allowing the bioprosthesis to be positioned completely superior to the annulus
- The internal diameter of the valve aligns with the patient’s annulus allowing for a larger available flow area
- The valve design allows blood to flow through the annulus encountering only tissue, not obstructive components such as the stent and sewing ring
SUITABILITY FOR FUTURE INTERVENTIONS
Valve dimensions and geometry enable future TAV-in-SAV replacements.
- Radiopaque annulus ring and stent post markers provide visible, distinct guidelines during TAV-in-SAV procedures
- MR conditional, non-metallic frame mitigates risk of corrosion between SAV and TAV stent materials
- Interior-mounted leaflets mitigate potential risk of coronary obstruction